- No products in the cart.
Menu
Start typing to see products you are looking for.
Start typing to see products you are looking for.
Browse Categories
-
PPE Apparel
Masks
-
 KN95 Respirator Mask, 4 layers, Box of 50 -LP-KN95-001BOX50
$79.00
KN95 Respirator Mask, 4 layers, Box of 50 -LP-KN95-001BOX50
$79.00
-
 Harley L-188 N95 Particulate Respirator Mask, Box of 20 - LP-L188-N95
$36.00
Harley L-188 N95 Particulate Respirator Mask, Box of 20 - LP-L188-N95
$36.00
-
 Lab Pro Mask KN95 (K-N95) Pack of 50 - LP-MASK2-K95 - 40% OFF
Lab Pro Mask KN95 (K-N95) Pack of 50 - LP-MASK2-K95 - 40% OFF
$125.00$75.00 -
 Lab Pro 3ply Earloop Disposable Mask (Non-Surgical) (Box of 50) - 84% OFF
Lab Pro 3ply Earloop Disposable Mask (Non-Surgical) (Box of 50) - 84% OFF
$57.00$9.00
-
-
Chemicals
-
Wipes
-
Microscopes and Lighting
-
Hand Tools
Hand Tools
-
 Excelta Scissors - Micro Self-Opening - Straight - SS - Blade Length .38" - 346B
$32.16
Excelta Scissors - Micro Self-Opening - Straight - SS - Blade Length .38" - 346B
$32.16
-
 Excelta Pliers - Forming Dog Leg - Carbon Steel - 907-89B
$320.73
Excelta Pliers - Forming Dog Leg - Carbon Steel - 907-89B
$320.73
-
 Excelta Brushes - Cleanroom Safe - Straight - SS/Statig09 Bristles - 213A-N
$17.58
Excelta Brushes - Cleanroom Safe - Straight - SS/Statig09 Bristles - 213A-N
$17.58
-
 Excelta Scissors - Micro Self-Opening - Straight - SS - Carbide Inserts - Blade Length .63" - 346-6-HT
$86.35
Excelta Scissors - Micro Self-Opening - Straight - SS - Carbide Inserts - Blade Length .63" - 346-6-HT
$86.35
-
-
Gloves
Powder Free Nitrile Gloves
-
 TrueForm Powder-Free Nitrile Exam Gloves Royal Blue (Small), Case of 1000 (TF-050-095-RB) (10 boxes 100/Box) - N301342
TrueForm Powder-Free Nitrile Exam Gloves Royal Blue (Small), Case of 1000 (TF-050-095-RB) (10 boxes 100/Box) - N301342
$199.00$99.00 -
 PowerForm Nitrile Exam Gloves Black (Medium), Case of 1000 (PF-90BK)(10 boxes 100/Box) - N716883
$237.00
PowerForm Nitrile Exam Gloves Black (Medium), Case of 1000 (PF-90BK)(10 boxes 100/Box) - N716883
$237.00
-
 TrueForm Powder-Free Nitrile Exam Gloves Royal Blue (Medium), Case of 1000 (TF-050-095-RB) (10 boxes 100/Box) - N301343
TrueForm Powder-Free Nitrile Exam Gloves Royal Blue (Medium), Case of 1000 (TF-050-095-RB) (10 boxes 100/Box) - N301343
$199.00$99.00
-
-
Swabs and Applicators
Foam Tipped Swabs
-
ESD & Static Control
ESD & Static Control
-
Lab Equipment
-
Pipettes
-
Furnaces and Ovens
Furnaces and ovens
-
All Products
- Services New
-
Industries Served
- Brands
-
Information
-
Blog
-
All blogs
- Aerospace
- Calibration of Lab Equipment
- Chemicals and Solvents
- Cleanroom and Critical Environment
- Electric Battery Labs
- ESD Safety
- Lab Consumables
- Lab Glassware and Glassware Equipment
- Lab Pro’s Top 5
- Laboratory Equipment
- Laboratory Safety & Lab Efficiency
- Medical Device Industry
- Microscopes, Lighting & Inspection
- News
- Our Blog
- Pipettes
- PPE and Safety Apparel
- Press Release
- Science Education
- Solar Energy Labs
- Sustainable & Eco-Conscious Lab
- Swabs
- Tweezers and Cutters
- Ultrasonic Cleaning
- VMI for Lab Supplies
-
All blogs
- Contact
Aldon
- Sort by
- FILTER BY PRICE
-
-
Below $100.00
-
$100.00-199.00
-
$200.00-299.00
-
$300.00-399.00
-
$400.00-499.00
-
Above $500.00
-
- FILTER BY COLOR
-
-
- FILTER BY SIZE
-
-
S
-
M
-
L
-
XL
-
2XL
-
3XL
-
- FILTER BY BRAND
-
-
Adidas
-
Camel
-
Motorola
-
Rolex
-
Samsung Galaxy
-
Seiko
-
Sony
-
Formation Of Esters Ap Chemistry Kit -IS8005
$59.99The yield of ester can be increased either by removing one of the products of the reaction as it is formed or by increasing the concentration of one of the...
Lechatelier's Principle -IS8004
$43.00Le Chatelier’s Principle is a qualitative rule, which allows the prediction of the effect of temperature, pressure and concentration changes on chemical reactions. The principle states: A chemical system at...
Mole Ratio Reactants Ap Chemistry Kit -IS8003
$39.75Using the method of continuous variation, two solutions are combined in various ratios. To select the ratio that produces the most product or consumes the most reactants, students must find...
Activity Series Ap Chemistry Kit -IS8002
$105.00An activity series of metals is a table of metals arranged in the order of their decreasing chemical activity or the ease at which the metal will give up one...
Thermochemistry & Hess Law Ap Chemistry Kit -IS8001
$45.00Employ Hess’s Law of enthalpy based on the observed data for two reactions. Students can measure the temperatures of the reaction in a calorimeter and analyze the data calculating the...
Electric Art -IS7049
$38.00A copper “pen” will be used to electrolyze a solution of potassium iodide and draw on a piece of filter paper. The color of the “ink” will change depending on...
Electrolysis Of Tin -IS7048
$21.95The electrolysis of a tin (II) chloride solution demonstrates the oxidation and reduction of Sn2+ ions as beautiful tin crystals grow and spread throughout the solution. There are enough materials...
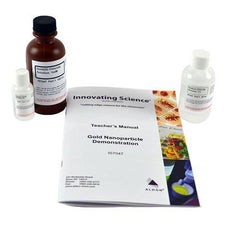
Gold Nanoparticle Demonstration -IS7047
$39.75Explore the interesting properties of nanomaterials by reducing a solution of gold (III) chloride to form colloidal gold nanoparticles. As the reaction occurs, the mixture undergoes multiple color changes before...
Rainbow Electrolysis -IS7046
$17.50Study oxidation-reduction, electrolysis and acid-base reactions as Universal Indicator is added to an aqueous sodium sulfate solution. When two carbon electrodes are connected to a battery and placed in the...
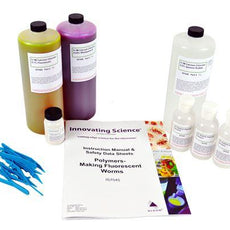
Polymers - Making Fluorescent Worms Chemistry Demonstration -IS7045
$45.00Allow students to learn about polymers and cross-linking using this fun and colorful activity. Students add a solution of sodium alginate to three calcium chloride solutions, each containing a different...
War Of The Acids -IS7044
$60.00Many people think that all acids are the same. Often times when students hear the phrases strong acid and weak acid they will confuse these concepts with high/low concentration of...
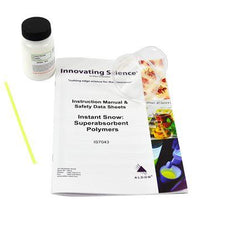
Snow Polymer Demonstration -IS7043
$16.50Demonstrate the wonderful, super absorbent properties of a polymer while making snow in your classroom. As the hydrophilic polymer instantly absorbs water, it will expand to over 40 times its...
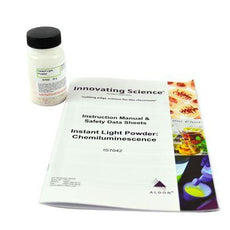
Instant Light Powder Kit -IS7042
$12.99In this demonstration, students will observe an example of a chemiluminescence reaction. Instant light powder and water are placed in a beaker producing a blue light. This demonstration is designed...
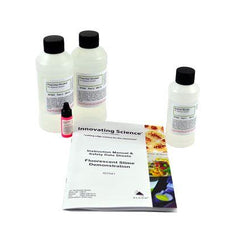
Fluorescent Slime Using Polyvinyl Alcohol -IS7041
$23.25Create your own fluorescent polymers (Slime) in this great classroom demonstration. Mixture is pink under regular light and glows orange under a black light. Included pigment will make this polymer...
Thionin And Iron: A Light Induced Redox Reaction -IS7040
$26.75In this lab a thionin-iron solution is prepared. Exposing the solution to light energy causes the colored thionin to be reduced by iron (II) ions, resulting in a clear solution....
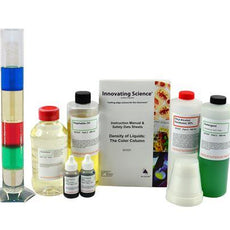
Density Of Liquids: The Color Column Demonstration Kit -IS7037
$62.00Several liquids of varying color are added to a cylinder. Due to differing densities of the liquids, a density gradient is formed resulting in a layered, multi-colored column. As an...
Forming Red, White & Blue Chemical Demonstration Kit -IS7036
$33.75Introduce chemical reaction types. This is a patriotic demonstration that illustrates complex ion formation, double replacement while introducing chemical reaction types.
Water To Wine Chemical Demonstration Kit -IS7035
$43.75Study acid base indicators and complex ions. Mixing three colored chemical solutions results in another wine colored solution. Adding the fourth chemical of sodium fluoride turns the solution clear.
Lechatelier's Principle -IS7034
$30.99Use an overhead projector to study the reaction of Le Chatelier’s principle.
Genie In A Bottle - Hydrogen Peroxide Decomposition Demo -IS7033
$34.99In this experiment you will show the decomposition of the chemical hydrogen peroxide using sodium iodide.
Formation Of Tin Wool Chemical Demonstration Kit -IS7032
$62.00This is a single replacement reaction in which the zinc metal dissolves in solution and tin metal is formed.
Oxidation-Reduction Of Complex Ions Chemical Demonstration -IS7030
$27.99Hydrogen peroxide is added to a solution of sodium potassium tartrate and heated. Copper sulfate solution is added and the solution turns light blue. With continued heating the solution foams...
Underwater Fireworks Chemical Demonstration Kit -IS7029
$24.50Calcium carbide is placed in a cylinder containing water. A reaction occurs producing acetylene gas bubbles. A plastic tube from a chlorine gas generator is placed in the cylinder. As...
Oxidation Of Glycerin Chem. Demo Kit USA -IS7028-AIR
$22.00A small amount of potassium permanganate is placed in a petri dish. A few drops of glycerin are added and after a few seconds, a puff of smoke and violet...
Oxidation Of Glycerin Chemical Demonstration Kit -IS7028
$21.00A small amount of potassium permanganate is placed in a petri dish. A few drops of glycerin are added and after a few seconds, a puff of smoke and violet...
Oscillating Reaction- The Redox Rainbow Chem Demo -IS7027
$24.75For this demonstration, add hydrogen peroxide to a colorless solution of potassium iodide to produce a red color, or you can add the hydrogen peroxide to a purple solution of...
The Oxidation-Reduction Flag Chemical Demonstration Kit -IS7026
$29.95You will be able to demonstrate the principles of a redox reaction. Paint the potassium thiocyanate solution and potassium ferrocyanide solution onto a filter paper. Once they are dry you...
Solid Fuel Chemical Demonstration Kit -IS7025
$24.99Two solutions are poured together in a beaker and form a gel. The gel can be ignited with a match. Commercially this gel is sold as Sterno®.
Volcano-Dehydration Of Sucrose Chemical Demonstration Kit -IS7021
$35.25Create your own volcano for a science fair demonstration.
Dehydration Of Sucrose Chemical Demonstration Kit -IS7020
$35.00Sulfuric acid is poured on sucrose. The resultant reaction produces a large amount of black foam as well as steam and smoke.
Polyurethane Foam Chemical Demonstration Kit -IS7018
$58.00Make your own foam and explain how when two viscous liquids are mixed together, they initiate a reaction producing a light weight polyurethane foam which expands to 30 times the...
Negative Coefficient Of Solub- Ility Chemical Demo -IS7016
$20.50Explain how there are exceptions to the rules of chemistry. Heat a solution and watch it form a precipitate. Watch it go back into solution as it cools.
Chemical Battery Chemical Demonstration Kit -IS7015
$29.99Construct a simple wet cell battery. When complete it will generate 1.5 volts for 20-30 minutes. Several cells can be connected in series to increase the voltage or in parallel...
Formation Of Eutectic Alloys Chemical Demonstration Kit -IS7014
$33.00Form a metal alloy as bismuth and tin are heated to the eutectic point of the two metals which is lower than the melting point of either metal.
Chemiluminescence Chemical Demonstration Kit -IS7013
$29.99Explain and view chemiluminescence. Observe how luminol is converted to an excited state in the presence of an oxidizer such as hydrogen peroxide. As the excited state molecule decays or...
Surface Tension Of Water Chemical Demonstration Kit -IS7012
$19.99Explain the principle of high surface tension as water acts like a membrane stretched across the surface preventing the sulfur particles from sinking. A wetting agent lowers the surface tension...
Formation Of Silicate(Red & White Precipitates Chemdemo -IS7011
$22.95Show how silicates of divalent ions are insoluble in water whereas silicates of monovalent ions are soluble.
Nylon (6-10) Rope Trick Chem. Demo Kit Air Version Usa -IS7010-AIR
$29.99Show an example of polymer chemistry. Make your own nylon which is a synthetic compound of high molecular weight that consists of up to millions of repeated chemical units that...
Nylon (6-10) Rope Trick Chemical Demonstration Kit -IS7010
$28.50Show an example of polymer chemistry. Make your own nylon which is a synthetic compound of high molecular weight that consists of up to millions of repeated chemical units that...
Thin Layer Chromatography Chemical Demonstration Kit -IS7009
$49.99TLC is a technique of separation chemistry, which provides a simple, rapid method of separating small amounts of compounds. A mixture of three dyes is prepared and then separated on...
Synthetic Rubber Chemical Demonstration Kit -IS7008
$36.00Show how the first synthetic rubber was made in United States by a reaction of sodium polysulfide with ethylene chloride. This produces a simple condensation polymer consisting of repeating units...
Blue Bottle Reaction Chemical Demonstration Kit -IS7007
$35.25Discover the principle of oxidation-reduction. Compounds that change color as a result of a change in their pH or oxidation state are called indicators. Illustrate this principle by mixing potassium...
Exothermic/Endothermic Rxns Chemical Demonstration Kit -IS7006
$43.50Study both processes of endothermic and exothermic reactions. Reactions are safely carried out in plastic bags. The students can pass the bags around to feel the results of the reactions.
Endothermic Reactions Chemical Demonstration Kit -IS7005
$25.00Study the process of endothermic reactions. The reaction is safely carried out in a plastic bag. The students can pass the bag around to feel the result of the reaction.