Lab Pro Glove Selection Guide
Glove Sizing Chart
Industrial and medical boxed gloves serve one purpose: to protect the operators. Cleanroom bagged gloves are designed to protect both the operators and the products. The primary purpose of wearing gloves in a controlled environment is to minimize submicron particle contamination of the product or process from the operator's hands. Therefore, choosing the most appropriate glove is one of the most important cleanroom consumable selections.
How to Select the Right Glove Size?

Picture A
To select your glove size, measure (in inches) around your palm indicated by picture A. Lab Pro indicates glove sizes by letter (XS, S, M, L, XL, XXL), use the table below to find the equivalent translation.
Choose the letter size from the chart that is the closest to the number you measured. For example: if your hand measures 10", then choose a size "L"
Choose the cuff length that you need

Picture B
To identify the correct glove length you need, measure from the top of the middle finger to where the cuff edge should be on forearm as shown in Picture B, then select the proper cuff and length.
Lab Pro offers three cuff lengths: 9", 9.5" and 12". The longer the length, the more added protection to your products.
| Glove Sizing Chart | ||
|---|---|---|
| Size | Inch | Centimeter |
| XS | 7" | 18 cm |
| S | 7½"-8" | 19-20 cm |
| M | 8½"-9" | 22-23 cm |
| L | 9½"-10" | 24-25 cm |
| XL | 10½"-11" | 26.5-28 cm |
| XXL | 11½"-12" | 29-30.5 cm |
Nitrile and Latex Comparison
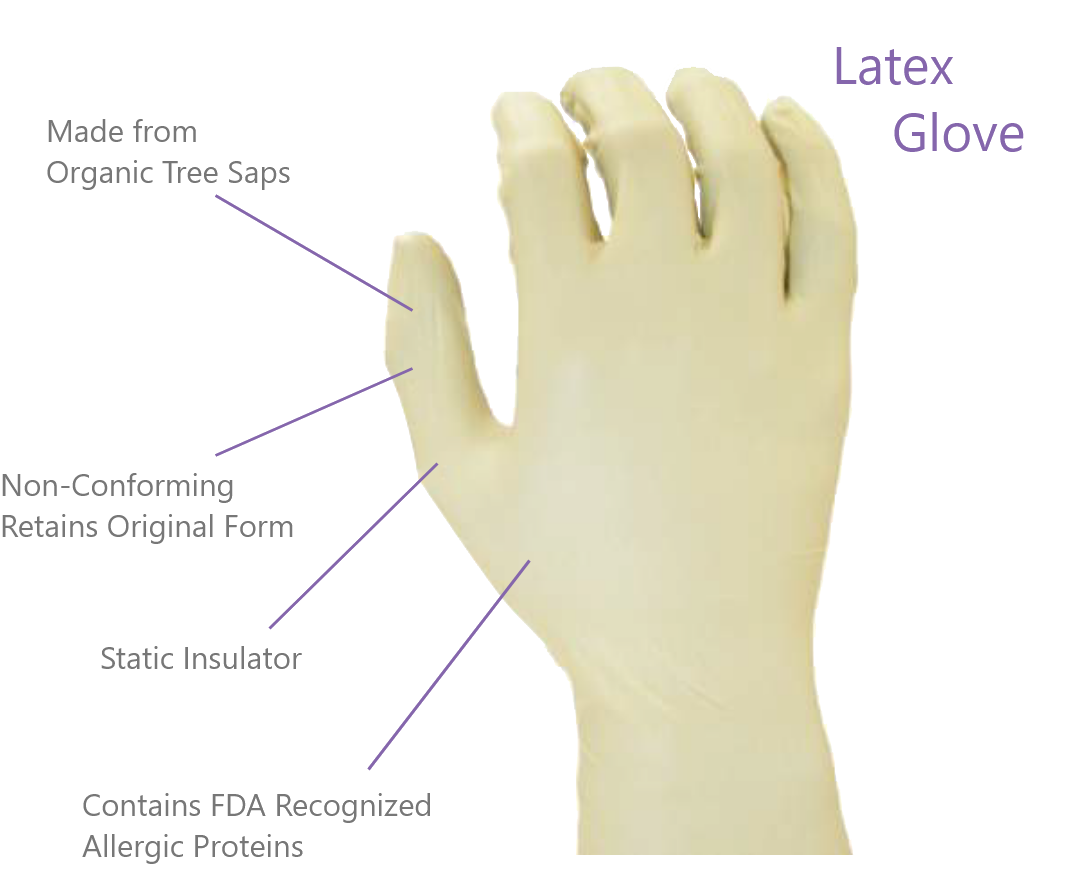
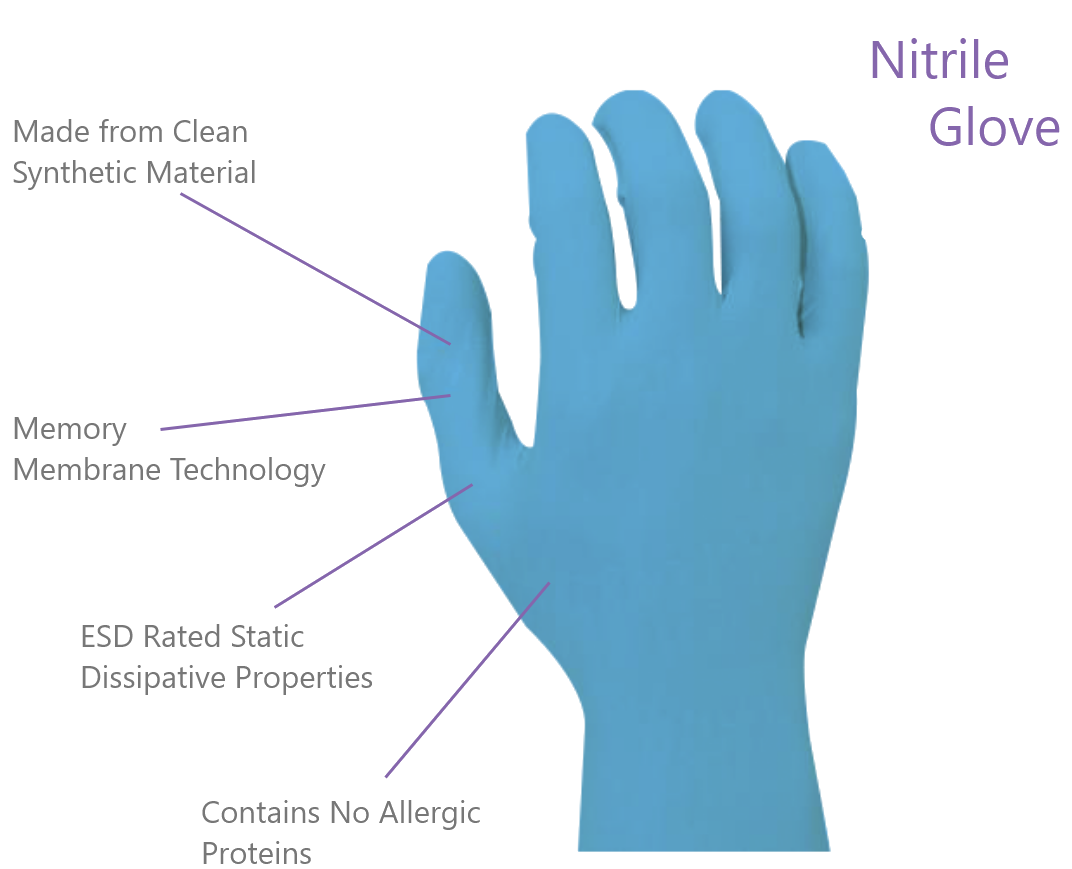
Powder free Nitrile gloves are the most commonly used clean and reliable option for laboratory and cleanroom operators. While Latex is made from organic tree sap and contains allergy-causing proteins recognized by the FDA, Nitrile is manufactured from a 100% inorganic synthetic material. Nitrile gloves provide superior barrier protection, minimize the risk of allergic reactions and possess excellent electrostatic discharge (ESD) properties. Nitrile with its memory-membrane is the most common option for operator comfort. This feature allows a nitrile glove to adapt to each individual's hand, improving motion and minimizing fatigue.
| Nitrile and Latex Characteristics Comparison | |||||
|---|---|---|---|---|---|
| Particle Count | Extractable Count | ESD Properties | Dexterity | Chemical Compatibility | |
| Nitrile | ++ | ++ | + | ++ | ++ |
| Latex | - | - | - | + | + |
Five Glove Selection Criteria

Particle Count
Numbers of particulate matter (contaminants) that comes off the gloves, typically measured in a dry test. Nitrile has the lowest amount of particles, followed by PVC, and latex has the highest particle count.

Electrostatic Discharge (ESD) Properties
The capacity of a glove to dissipate, or conduct a static charge to a ground. Both PVC and nitrile exhibit excellent ESD qualities. Latex is insulative.

Chemical Compatibility
Latex is a good choice for protection against acids. Nitrile has better resistance than latex to a broad range of chemicals and performs well with solvents and acids. PVC has poor acid and solvent based resistance and is best suited for a dry environment. Note: Thin-walled gloves are only intended for splash protection.

Extractable Count
The amount of elements extracted from the gloves in an immersion test, generally measured in Parts Per Billion (PPB) or Grams per Square Meter (g/m2). 18 mega ohm DI H2O washed nitrile has the lowest ionic counts, followed by PVC and latex.

Dexterity
Latex gloves fit tight on the hand, but have no "memory" to conform to a specific hand shape. Nitrile's memory (modulus) properties provide more comfort, but are lower in elasticity than latex. PVC has much lower dexterity and is recommended when tactile requirements are low.
The Only Guide You'll Need to Choosing The Right Gloves
The primary purpose of wearing gloves in a controlled environment is to minimize sub-micron particle contamination of the product or process from the user's hands. Therefore, choosing the most appropriate glove is one of the most significant cleanroom consumable selections. To help you get started, download Lab Pro's selection guide that covers the following topics:
- Glove Substrate Selection Chart
- Glove Sizes
- Glove Chemical Compatibility
- Nitrile and Latex Comparison
Get the job done safely and efficiently with the right protective gloves for your work. Download our guide now!
Get My Guide NowNitrile and Latex Comparison
-
Latex Gloves (Natural Rubber)
Known for its durability, comfort, and dexterity, Latex gloves are the most common material for gloves. Latex is ideal when high dexterity and tactile sensitivity is required. It provides excellent protection against contamination and chemicals such as alcohols, acids, and ketones. In some people, latex proteins may cause an allergic reaction for which nitrile gloves provide a good alternative.
-
Nitrile Gloves (Acrylonitrile Butadiene Rubber)
Nitrile is the most common material for gloves used in for laboratory and cleanroom environments. Nitrile Gloves offers good dexterity and superior resistance to many types of chemicals. Compared to latex, nitrile is lower in particles, ionic extractable and non-volatile residues. Nitrile also has "memory" properties that adapt it to the hand for a more custom fit that reduces fatigue. It is three times more puncture resistant than latex, and it also provides protection against cutting and scraping. Static-dissipative properties make nitrile ideal for use in medical device manufacturing, microelectronics, and other similar dry controlled environments.
-
Vinyl Gloves (PVC - Polyvinylchloride)
PVC Gloves better known as Vinyl is proven for its ESD properties, inherent cleanliness (low in both particles and ionic extractable), and low cost. These attributes make vinyl gloves ideal for use in medical, restaurant and hospitality industry. A PVC glove fit looser on an operator's hand than latex or nitrile, which can reduce dexterity.
Cleanroom Gloves

Cleanroom gloves are bagged and designed to protect both the operators and the products. The primary purpose of wearing gloves in a controlled environment is to minimize sub micron particle contamination of the product or process from the operator's hands. Therefore, choosing the most appropriate glove is one of the most important cleanroom consumable selections. Ask a Lab Pro specialist how we can help you find the best Cleanroom glove for your laboratory needs.
| Glove Substrate Selection Chart | |||||
|---|---|---|---|---|---|
| Vinyl | Latex | Nitrile | |||
| Static Dissipative | ++ | ++ | ++ | ||
| Protein Allergies | - | - | - | ||
| Chemical Resistance | - | ++ | +++ | ||
| Strength/Durability | + | +++ | ++ | ||
| Modulus | - | ++ | +++ | ||
| Tactile Sensitivity | - | ++ | +++ | ||
| - Poor + Good ++ Better +++ Best | |||||
Glove Chemical Compatibility Chart
| Chemical | Nitrile | Latex | Vinyl |
|---|---|---|---|
| Acetaldehyde | |||
| Acetone | |||
| Acetic Acid | |||
| Acetylene Gas | |||
| Aluminium Sulfate | |||
| Ammonium Hydroxide, Dilute | |||
| Ammonium Nitrate | |||
| Ammonium Sulfate | |||
| Amyl Alcohol | |||
| Anline | |||
| Anline Oil | |||
| Animal Fats | |||
| Animal Oils | |||
| Antifreeze | |||
| Barium Sulfite | |||
| Beet Sugar Liquors | |||
| Benzyl Alcohol | |||
| Borax | |||
| Boric Acid | |||
| Brake Fluid | |||
| Butyl Alcohol | |||
| Butyle Cellusolve | |||
| Calcium Bisulfite | |||
| Calcium Chloride | |||
| Calcium Disulfide | |||
| Calcium Hydroxide | |||
| Calcium Hypochlorite | |||
| Carbolic Acid | |||
| Carbon Dioxide | |||
| Castor Oil | |||
| Caustic Potash | |||
| Caustic Soda | |||
| Chlorine Solution | |||
| Chromic Acid 30% | |||
| Citric Acid | |||
| Copper Chloride |
| Chemical | Nitrile | Latex | Vinyl |
|---|---|---|---|
| Copper Sulfate | |||
| Cotton Seed Oil | |||
| Cresol | |||
| Cupric Nitrate | |||
| Cyclohexane | |||
| Cyclohexanol | |||
| Dibutylphthalate | |||
| Dietylether | |||
| Di-n-amylamine | |||
| Di-n-butymine | |||
| Di-n-butyl Phthalate | |||
| Di-n-octyle Phthalate | |||
| Diallylamine | |||
| Diesel Fuel | |||
| Diethanolamine | |||
| Diethylamine | |||
| Dimethylsulphoxide | |||
| Diisobutyl Ketone | |||
| Diisobutyllamine | |||
| Dimethyl Ether | |||
| Dimethyl Sulfoxide | |||
| Epoxy Resins, Dry | |||
| Ethane Gas | |||
| Ethanol | |||
| Ethyl Acetate | |||
| Ethyl Alcohol | |||
| Ethyl Ether | |||
| Ethylene Glycol | |||
| Fatty Acids | |||
| Ferric Chloride | |||
| Ferrous Sulfate | |||
| Formaldehyde | |||
| Formic Acid | |||
| Freon 113 Or Tf | |||
| Gasoline, 40-50% | |||
| Glucose |
| Chemical | Nitrile | Latex | Vinyl |
|---|---|---|---|
| Glutaraldehyde,<5% | |||
| Glycerine | |||
| Glycerol | |||
| Glycol | |||
| Grain Alcohol | |||
| Heptanes | |||
| Hexamthyldisiloxane | |||
| Hexane | |||
| Hydrazine | |||
| Hydrochloric Acid, Dilute | |||
| Hydroflouric Acid, Dilute | |||
| Hypochlorites | |||
| Hydrogen Gas | |||
| Hydrogen Peroxide | |||
| Hydrogen Sulfide | |||
| Inorganic Salts | |||
| Iron Chloride | |||
| Iron Sulfates | |||
| Isobutyl Alcohol | |||
| Isooctane | |||
| Isopropanol | |||
| Jet Fuel, <30% Aromatics | |||
| 2- Propanol | |||
| Kerosene | |||
| Lactic Acid | |||
| Lauric Acid | |||
| Lineoleic Acid | |||
| Magnesium Chloride | |||
| Magnesium Sulfate | |||
| Malathion, 30-70% | |||
| Maleic Acid | |||
| Methanol | |||
| Methyl Ethyl Ketone | |||
| Methyl Isobutyl Ketone | |||
| Mercuric Chloride | |||
| Mercury |
| Chemical | Nitrile | Latex | Vinyl |
|---|---|---|---|
| Moisture | |||
| Monoethanolamine | |||
| Muriatic Acid | |||
| N-Butyl Alcohol | |||
| N- Methyl-2-Pyrrolidone | |||
| N-Propyl Alcohol | |||
| Naptha, 15-20% Aromatics | |||
| Naphthalene | |||
| Nitric Acid <30% | |||
| Nitrobenzene | |||
| Octane | |||
| Octyl Alcohol | |||
| Oleic Acid | |||
| Oxalic Acid | |||
| Palmitic Acid | |||
| Pentachlorophenol | |||
| Pentane | |||
| Perchloric Acid, 30-70% | |||
| Phenol | |||
| Phosphoric Acid | |||
| Pickling Solution | |||
| Picric Acid | |||
| Pine Oil | |||
| Potash Salts | |||
| Potassium Bromide | |||
| Potassium Carbonate |
| Chemical | Nitrile | Latex | Vinyl |
|---|---|---|---|
| Potassium Chloride | |||
| Potassium Cyanide | |||
| Potassium Dichromate | |||
| Potassium Ferrocyanide | |||
| Potassium Hydroxide | |||
| Potassium Hypochlorite | |||
| Potassium Iodide | |||
| Potassium Nitrite | |||
| Potassium Phosphate | |||
| Potassium Silicate | |||
| Potassium Sulfate | |||
| Potassium Sulfide | |||
| Potassium Thiosulfate | |||
| Propan-2-Ol | |||
| Propyl Alcohol | |||
| Propylene Glycol | |||
| Rust Inhibitors | |||
| Rock Salt | |||
| Salt Spray | |||
| Sodium Acetate | |||
| Sodium Azide | |||
| Sodium Bicarbonate | |||
| Sodium Carbonate | |||
| Sodium Chloride | |||
| Sodium Cyanide | |||
| Sodium Hydroxide |
| Chemical | Nitrile | Latex | Vinyl |
|---|---|---|---|
| Sodium Hypochorite | |||
| Sodium Nitrate | |||
| Sodium Phosphate | |||
| Sodium Silicate | |||
| Sodium Sulfate | |||
| Sodium Sulphite | |||
| Sodium Thiosulfate | |||
| Stearic Acid | |||
| Sulfur Dioxide | |||
| Sulfuric Acid (Dilute) | |||
| Sulfurous Acid (75-100%) | |||
| Tannic Acid | |||
| 1,2,4,5- Tetrachlorobenzen | |||
| Tetrahydrofuran | |||
| Triethanolamine | |||
| Turpentine | |||
| Vegetable Oils | |||
| Water (Soapy) | |||
| Water (Distilled) | |||
| Water (Fresh) | |||
| Water (Salt) | |||
| Wood Alcohol | |||
| Xylene | |||
| Zinc Salts |
*Please note: The data listed on our compatibility chart should be used as a reference guide as they are recommendations only. Always refer to the chemical Safety Data Sheet(SDS) and warning labels when handling any of these chemicals. The chemicals mentioned above are meant to be used for research,industrial work, cleaning or disinfecting and should always be stored out of the reach of young children or infants.
| Resistance |
Permeation
1ug/cm2/second |
|
|---|---|---|
| Excellent | > 300 | |
| Good | 181 - 300 | |
| Fair | 61 - 180 | |
| Poor | 20 - 60 | |
| Not Recommended | < 20 |




